
API's
Active Pharmaceutical Ingredients

Active Pharmaceutical Ingredients
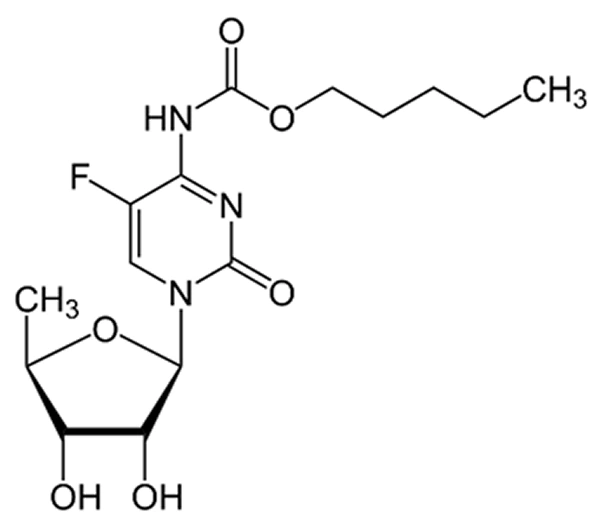
Active Pharmaceutical Ingredient (API)
USP / BP / EP (as applicable)
Capecitabine
C₁₅H₂₂FN₃O₆
~359.35 g/mol
154361-50-9
5-Deoxy-5-fluoro-N-[(pentyloxy)carbonyl]-cytidine
Fluoropyrimidine carbamate (prodrug of 5-FU)
Antineoplastic / Anticancer Agent
| Appearance | White to off-white crystalline powder |
|---|---|
| Solubility | Poorly soluble in water but increasing in polar organic solvents like methanol, ethanol, DMSO, ethyl acetate |
| Melting Point | 110-121°C |
| pH | 1.3 to 1.7 |
Capecitabine is an orally administered prodrug of 5-fluorouracil (5-FU). It is designed to be selectively activated in tumor tissues, thereby reducing systemic toxicity while delivering cytotoxic activity against rapidly dividing cancer cells. It is widely used in the treatment of colorectal, breast, and gastric cancers.
Capecitabine is converted in the body into 5-fluorouracil (5-FU), a pyrimidine analog that inhibits thymidylate synthase, thereby blocking DNA synthesis and repair. This leads to cell cycle arrest and triggers apoptosis in rapidly dividing tumor cells. Because Capecitabine is selectively activated in tumor tissues, it helps reduce systemic side effects compared to direct 5-FU injections.
| Attribute | Why Salius Pharma Is a Good Choice |
|---|---|
| Quality Certification | WHO GMP, ISO 9001:2015, FDA audited facilities |
| Affordability & Generic Access | Cost competitive generics for global supply |
| Export Experience | Proven track record of supplying globally |
When you order from Salius Pharma, you receive more than just a product — you receive a complete, professionally prepared package that meets international regulatory and quality expectations.
We ensure prompt and reliable shipping worldwide. Delivery times vary depending on destination, order volume, and regulatory requirements.
7 to 21 business days from dispatch, depending on your location.
Capecitabine is an oral prodrug that is enzymatically converted in tumor tissues to 5-fluorouracil (5-FU). The active metabolite inhibits thymidylate synthase, leading to impaired DNA synthesis and inducing apoptosis in rapidly dividing cancer cells.
Capecitabine is used in the treatment of colorectal cancer, gastric cancer, and breast cancer, either as monotherapy or in combination with other anticancer agents, particularly where oral chemotherapy is preferred.
Capecitabine API can be manufactured to meet USP, EP, and BP specifications.
Export supplies are usually supported with batch-specific CoA, MSDS, GMP compliance documents, stability data, and regulatory dossiers such as DMF/CTD modules suitable for regulated and semi-regulated markets (US, EU, LATAM, MENA, and Africa).
Looking to source Capecitabine or other high-quality pharmaceutical products?
We’re here to help.
Whether you need Active Pharmaceutical Ingredients (APIs), finished formulations, or regulatory support,
our team is ready to provide dependable, compliant, and cost-effective solutions tailored to your market needs.
The contents of this page and any attachments are intended solely for the designated recipient(s) and may contain confidential and/or privileged information protected by law.
Any patented products are excluded from our offerings in regions where such patents are currently in force.